Neurological disorders – Alzheimer’s disease, Parkinson’s disease, and dementia – are on track to become the second leading cause of death in the United States. More than 20 million Americans are at risk of developing a debilitating neurodegenerative disease by 2050, and yet diagnostics and treatments are extremely limited.
“Not to be doom and gloom, but there are no good therapies out there right now,” said Julie Moreno, assistant professor in the department of environmental and radiological health sciences at Colorado State University. Moreno, who was recently named a 2021 Boettcher Investigator, conducts laboratory research in worms (C. elegans), mice, and dogs with the goal of developing diagnostics and treatments for neurodegenerative diseases in humans.
Common disorders like Alzheimer’s and Parkinson’s and rarer disorders such as Creutzfeldt-Jakob disease and Huntington disease all fall under the umbrella of protein-misfolding diseases, which begin with the accumulation of incorrectly-formed proteins in the brain. The toxic proteins trigger inflammation in the glial cells, which support and protect neurons, resulting in neuronal dysfunction and death.
“In all of these diseases the progression of disease is similar,” Moreno said. “The clinical signs differ due to the part of the brain that is first affected. Parkinson’s disease affects a part of the brain that controls your motor system, and Alzheimer’s disease affects a part of the brain that controls learning and memory.”
June is Alzheimer’s and Brain Awareness Month
Each June, the Alzheimer’s Association raises awareness about dementia and shows support for those living with the disease and their caregivers. 50 million people live with the disease. As that number grows, Colorado State University researchers and clinicians seek to understand the evolution of neurodegenerative diseases and develop diagnostics and treatments.
Early detection is essential
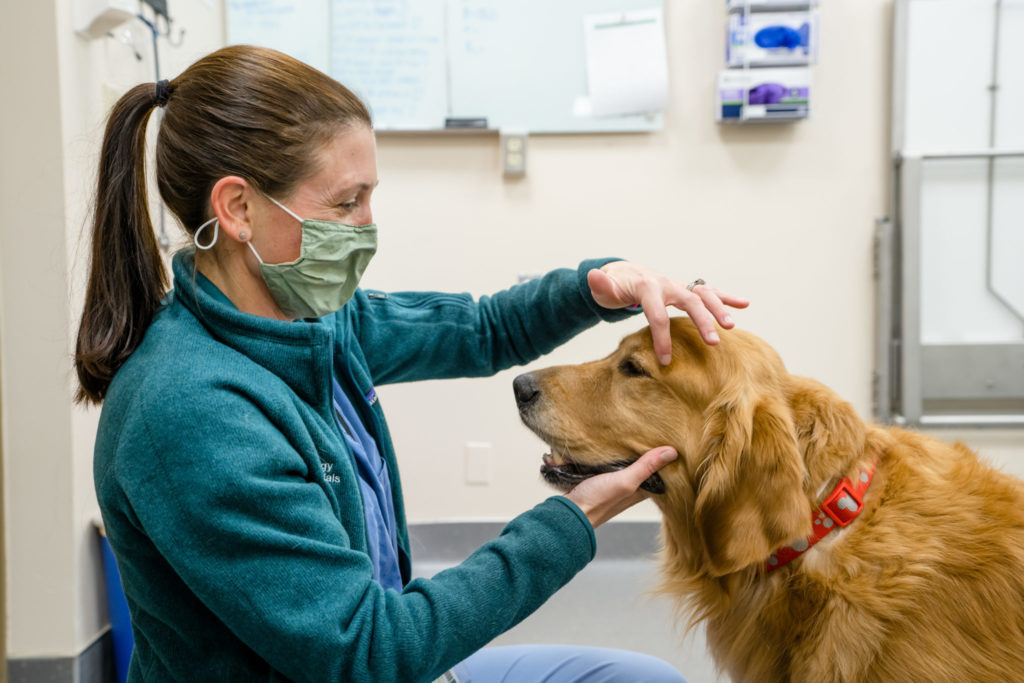
If your dog is getting up there in age, it’s important to be on the lookout for signs of canine cognitive dysfunction syndrome and see your veterinarian if you notice any changes. Early recognition of abnormal behavior is essential because diseases like canine cognitive dysfunction syndrome are degenerative, so as they progress, the worse they get, and deterioration of the brain is irreversible.
“Oftentimes dogs are diagnosed once they’ve shown a lot of clinical signs, but the ideal time to intervene is earlier on,” said Dr. Lisa Bartner, veterinary neurologist at the James L. Voss Veterinary Teaching Hospital.
If clinical signs are recognized, an MRI can help with diagnosis, and while there’s no cure for cognitive dysfunction, some physical therapy and rehabilitation techniques show promise in helping dogs maintain quality of life for longer.
Dogs and humans share environment, risk for dementia
A common misconception holds that genetics are the primary trigger for neurodegenerative diseases, yet only about 10% of cases are hereditary. The most significant risk factor for protein-misfolding diseases is aging. Because aging, stress, and dementia are common to many species, these diseases can be understood through translational research, which translates laboratory and preclinical research from one species to another, such as canines to humans. Moreno and veterinary neurologist Dr. Stephanie McGrath are studying canine cognitive dysfunction syndrome or “doggie dementia,” which affects 35% of canines over 8 years old.
As a companion species, dogs share everything about their environment—from sleep to food to exercise—with their humans, and therefore offer an ideal opportunity to study neurodegenerative diseases. With funding from CSU’s Translational Medicine Institute, Moreno and McGrath are studying the disease evolution and attempting to develop an early, non-invasive diagnostic tool that could be translated to human medicine.
“We’re looking at etiology and biomarkers for this disease, but it’s also broader than that,” McGrath said. “We’re looking at how aging in cognitively impaired dogs compares with human aging, and then how healthy aging compares with cognitive decline.”
Of mice and models
Moreno will use the Boettcher Foundation award to support translational research for therapies that can realistically advance from the laboratory to the clinic.
“The animal model we use in the science is key to how understanding how these therapies might work,” Moreno said. Mice are an ideal laboratory model because prion disease occurs naturally in mice with same presentation as in humans. Moreno hypothesizes that adult stem cells derived from fat tissues can reduce inflammation early in the disease.
“In a cell culture dish, we’ve been able to show that these stem cells secrete protective factors that kind of soak up all that inflammation in the brain,” Moreno said.
In later stages of the disease, Moreno will use stem cells from the nose tissues of the mice to replace lost neurons. This two-step treatment may alleviate the disease and increase the lifespan of the mice. If successful, it will present a new treatment model for neurodegenerative diseases in humans.
