
Two Colorado State University veterinarians are launching a pilot study of a novel spinal surgery to improve the success of the procedure, minimize post-operative discomfort, and decrease complications for paralyzed dogs.
Over the past two decades, human medicine made huge advancements in improving outcomes of spinal surgery through the use of minimally invasive surgery techniques. In contrast, veterinary spinal surgery has largely remained the same since it was first performed in the 1950s. As pets are an integral part of the nuclear family and owners aim to improve their pets’ longevity and quality of life, evolution of veterinary spinal surgical techniques should follow suit.

Dr. Eric Monnet and Dr. Lisa Bartner, both board-certified surgeons at the Colorado State University Veterinary Teaching Hospital, are at the forefront of this evolution with a study to evaluate the use of a minimally invasive surgical technique called an integrated endoscopic system to perform hemilaminectomies in short-legged dog breeds with thoracolumbar disc extrusions.
Eight dogs who meet qualifying criteria will be enrolled and randomly assigned to either open or minimally invasive surgery for the hemilaminectomy. The same protocol for all dogs will be followed for antibiotics, pain management, post-operative rehabilitation, and hospitalization time.

The dogs will be regularly evaluated for pain to determine if the minimally invasive approach is less painful than the open surgical approach. Dogs in both groups will also have neurologic examinations performed daily to monitor post-surgical improvements. Muscle injury will be compared between the two groups through bloodwork and magnetic resonance imaging (MRI) to assess whether minimally invasive surgery is less traumatizing.
This is the first study to evaluate the efficacy of minimally invasive surgery in paralyzed dogs. Monnet and Bartner hope the research will lead to improved surgical outcomes in dogs with disc protrusions.
Qualifying criteria
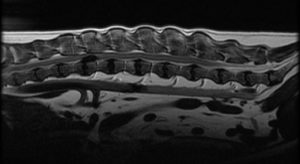 Acute (typically within 48-72 hours) total paralysis in both hind limbs
Acute (typically within 48-72 hours) total paralysis in both hind limbs- Deep pain present or, if absent, lost within 24 hours of initial presentation to CSU
- Weighs less than 15kg (roughly 32lbs)
- Chondrodystrophic breed (see list below)
- T3-L3 myelopathy
- No previous spinal problems
Because it is critical to do the surgery as soon as possible after a disc extrusion occurs, owners are advised to come to the Veterinary Teaching Hospital through the emergency and urgent care service so they can be seen immediately. Once initial qualification criteria are confirmed, the patient will be transferred to the neurology clinical trials team for enrollment.
Benefits of participating in this trial
- $2,500 credit toward total cost
- Discounted fees, including pre- and post-operative MRI scans, exam, X-rays
- If randomly selected for minimally invasive surgery, patient may experience less post-operative pain and faster recovery times
- Helping to advance surgical techniques
Common chondrodystrophic breeds
| American Cocker Spaniel | Clumber Spaniel | Pekingese |
| Australian Shepherd | Coton de Tulear | Pembroke Welsh Corgi |
| Basset Hound | Dachshund | Poodle (Miniature & Toy) |
| Beagle | Dandie Dinmont Terrier | Poodle (Standard) |
| Bichon Frise | English Springer Spaniel | Portuguese Water Dog |
| Boykin Spaniel | French Bulldog | Pug |
| Bulldog | German Hound | Rat Terrier |
| Cardigan Welsh Corgi | Havanese | Scottish Terrier |
| Cavalier King Charles Spaniel | Jack Russell Terrier | Shih Tzu |
| Chesapeake Bay Retriever | Lhasa Apso | Skye Terrier |
| Chihuahua | Maltese | Yorkshire Terrier |
| Chinese Crested | Nova Scotia Duck Tolling Retriever |
(Adapted from Chondrodystrophy (CDDY and IVDD Risk) and Chondrodysplasia (CDPA), UCDavis, and Animal Genetics)
For more information about this trial, please contact the Neurology Clinical Trials Coordinator Breonna Thomas by email at CSUNeuroTrials@colostate.edu or phone at (970) 297-4405.